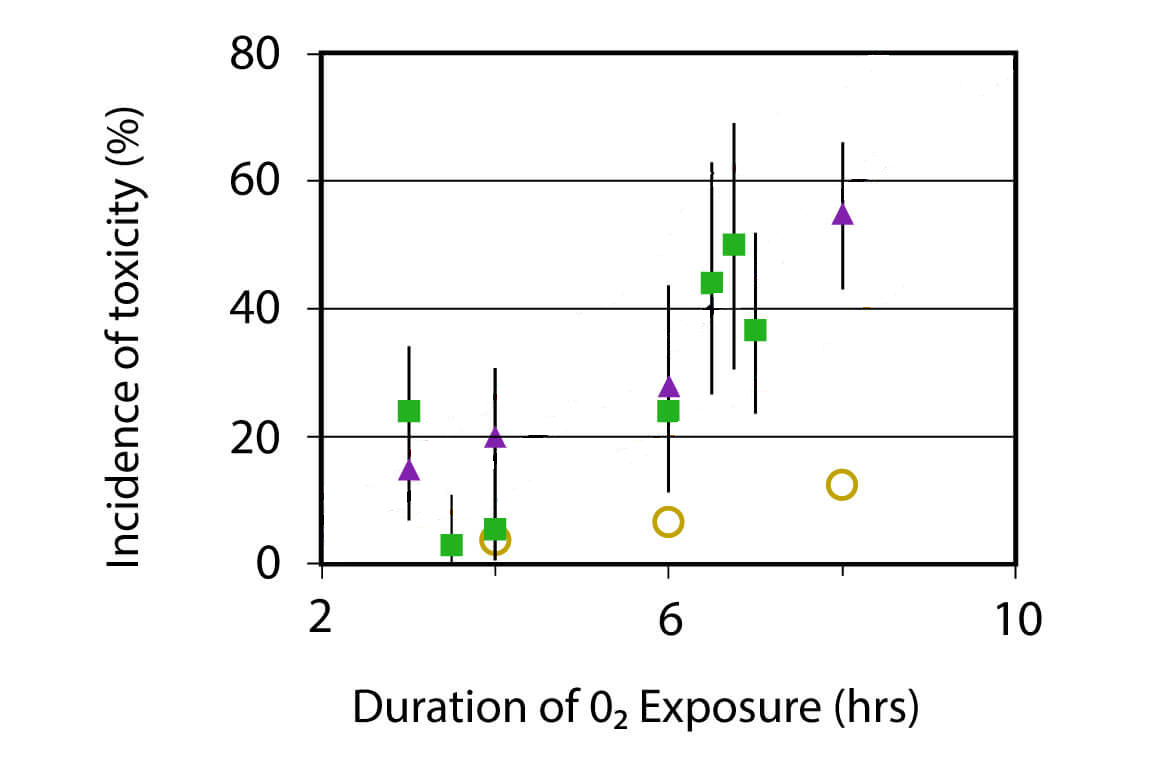
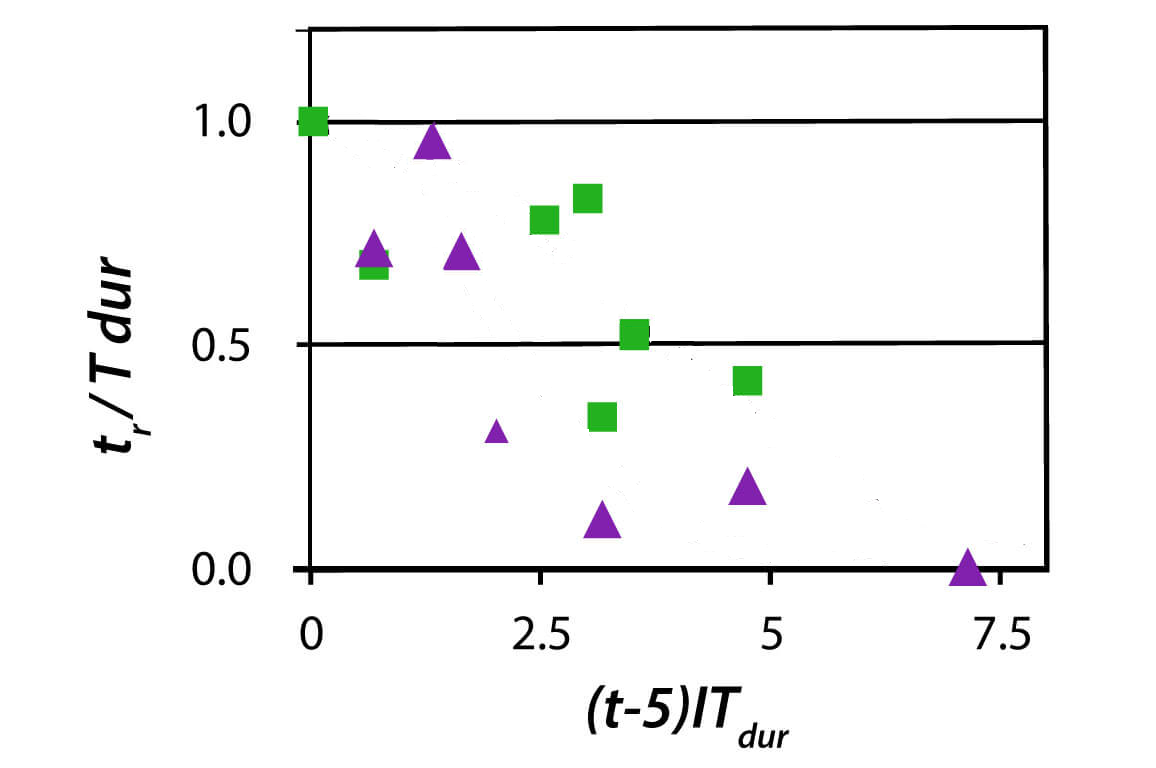
In the past, I have talked about the poor empirical data underlying the NOAA limits for preventing CNS oxygen toxicity. Of course, those are tricky, since having a seizure at depth has a high likelihood of resulting in fatal outcomes while there are good chances that the diver has no way to see it coming.
On the other hand, the limits have been quite restrictive: At a pO2 of 1.3 bar, you were supposed not to exceed 180 minutes in a single dive and 210 minutes in a 24 hour period. Sticking to such a limit (with 1.3 bar being a typical rebreather set-point) will make certain dives impossible as the decompression requirements exceed three hours. This had the consequence that many technical divers have been ignoring the limits coming from this “CNS clock”. Turns out, this has not led to many dead rebreather divers due to oxygen seizures at depth.
Turning this around, there is now more data to justify relaxing those limits while controlling the risk. In March 2025, at the American Academy of Underwater Sciences Annual Meeting, there was an expert workshop that resulted in new recommendations authored by Joseph Hoyt, F Gregory Murphy, Neal W Pollock, Dawn Kernagis, Nicholas Bird, Michael Menduno, John Bright, and Simon J Mitchell. They are still very conservative with their recommendations. For example, they only recommend new limits for pO2 of 1.3 bar although mentioning that it is highly likely that those times considered save would also be save at lower partial pressure. They make no statement about higher partial pressures like 1.4 bar or even 1.6 bar quoting insufficient controlled dive date in these ranges.
So, for 1.3 bar, they say, four hours of exposure during the “working part” of the dive plus an additional four hours during decompression at rest can be considered save.
They note that this is only about CNS toxicity as this has more potentially harmful consequences and pulmonary toxicity might have to be managed in addition. Furthermore they recommend considering additional safety measure like O2 breaks, breathing low density gases and using full face masks or habitats to mitigate consequences of a seizure.
PS: A few days ago, I had to learn that Michael Menduno, one of the authors of the paper mentioned above and editor of inDepth magazine has passed away. In the limited interactions I had with him he surprised me with is generous and welcoming attitude. Rest in peace!